COOLING SYSTEM
-
CONSTRUCTION
-
The FC cooling water ion exchanger assembly is installed to the FC cooling system, and when FC stack coolant passes through its internal FC cooling water ion exchanger element, ionic impurities in the FC stack coolant are removed, preventing degradation of the electrical generating efficiency of the FC stack.
Tech Tips
-
The higher the concentration of ionic impurities in the FC stack coolant, the higher its electrical conductivity becomes, and so the electricity generated in the FC stack flows into the FC stack coolant, resulting in degradation of electrical generating efficiency.
-
If the vehicle is left for a long time without being used, there is a possibility that the ionic impurities in the FC stack coolant will increase and electrical conductivity will become high. To restore the values to the normal range as quickly as possible, when the vehicle is first started after being left for a long time, the FC stack coolant flow rate will be increased to a higher rate than usual. Also, when the vehicle is first started after the auxiliary battery has been disconnected, the FC stack coolant flow rate will be increased because the system does not know how long the vehicle has been left unused for.
-
If the FC cooling water ion exchanger element is used beyond its limit, its ion exchange capabilities will degrade, and the insulation resistance of the FC stack coolant will be unable to be maintained. For this reason, when the replacement interval is near, a message will be displayed in the multi-information display of the combination meter prompting the user to replace the element.
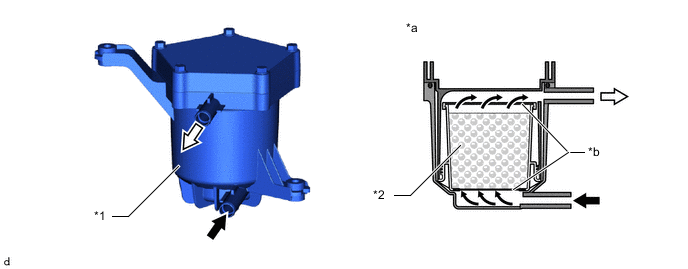
*1 FC Cooling Water Ion Exchanger Assembly *2 Ion Exchange Resin *a Representation of FC Cooling Water Ion Exchanger Assembly Flow Pathway *b Mesh 
Inlet 
Outlet -
The FC cooling water ion exchanger element uses a network-structure polymer substrate consisting of a styrene substrate with divinylbenzene as a crosslinking agent, and containing 2 types of ion exchange groups. The anion exchange resin uses quaternary ammonium groups as the exchange groups, and the cation exchange resin uses sulfonic acid groups as the exchange groups. The anion exchange groups accept electrons and so absorb negatively-charged anions, while the cation exchange groups give up electrons and so absorb positively-charged cations.
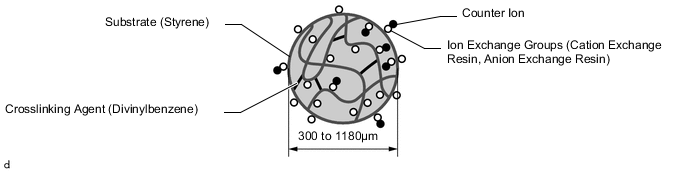
-
-